Week 6
Task 1 - Demonstration, Observations and the concept of measuring pH
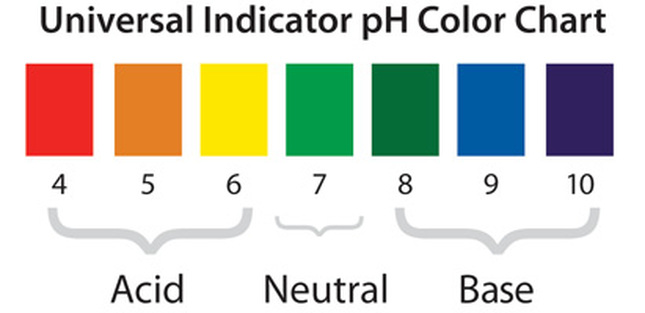
What is pH and how can we find the pH of a liquid or a solution?
In the test tube rack there are three test tubes containing liquid.
|
1. Describe the liquid in each test tube. Ask some questions that would help you make good observations.
2. Based on your observations, make some inferences about what the liquids could be. Ask some questions that would help you make good inferences. 3. Add 10 drops of Universal Indicator to each test tube. 4. Describe the change in each test tube. 5. Make some inferences about why the liquids are different colours. (Hint: did we add the same Universal indicator to each test tube?) |
UNIVERSAL INDICATOR AND THE pH SCALE
|
Task 2 - Investigation
Investigating the pH of aqueous solutions
Aim
Testing flocculants and other aqueous solutions to find out if they are acidic, neutral or basic.
You are given a collection of solutions. Some solutions are the flocculants that you have used in class. Some some solutions may be new to you, but you have probably eaten some of them before!
Note that in the laboratory we do not eat or taste anything!
Safety
During your Investigation you are to follow safe laboratory procedures.
1. Keep your work station tidy and dry.
2. Remain at your work station.
3. Replace the lid of each bottle immediately after using the bottle.
4. Hold the droppers above the test tube to drip the solution.
5. On completion of the Investigation return all apparatus and solutions.
Your Challenge.
Start by testing the pH of tap water and recording the colour.
You are to test each solution for its pH, using Universal Indicator.
Record your observations using the results table below.
Results Table for pH of solutions.
Analysing the Results
Build a table with three columns.
Write three headings, one in each column;
acidic solutions neutral solutions basic solutions
pH 1 to 6.5 pH 6.5 to 7.5 pH 7.5 to 14
Classify each of the solutions as either acidic solutions, neutral solutions, or basic solutions.
Conclusion
Give the pH number range for acidic solutions, neutral solutions and basic solutions.
Task 3 Application of your science understanding to the water cycle.
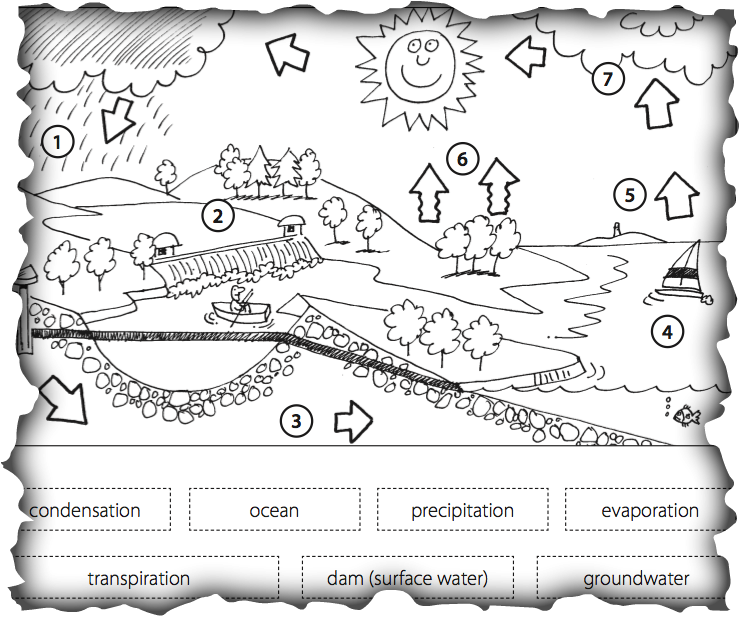
The Water Cycle
How does pH relate to the water cycle?
Based on your observations of pH, note on the water cycle the possible pH of different water bodies.
Beneath the diagram elaborate on your decisions.
Based on your understanding of solutions and changes of state you can predict the pH of the following types of water outlined in the water cycle.
Justify your answer.
Justify your answer.
- pH of rain water
- pH of dam water
- pH of fertilizers
- pH of ground water
- pH of ocean water
- pH of evaporated water
- pH of transpiration water
Task 4 - Testing some ideas
Distillation
|
Aim What is the pH of distilled water? Prediction Write your prediction. Explain why you think this. Equipment for distillation Distillation tube and delivery tube Collection tube Bunsen burner Protective mat matches Method and Diagram
|
Turn off the bunsen burner before you collect the Universal Indicator. Why?
Add three or four drops of Universal Indicator to the distilled water.
Results
Record the colour and the pH number.
Conclusion
Classify the distilled water as acidic, neutral or basic.
Explain why this is so.
Add three or four drops of Universal Indicator to the distilled water.
Results
Record the colour and the pH number.
Conclusion
Classify the distilled water as acidic, neutral or basic.
Explain why this is so.
Water Review for the Test
|
| ||||||