Week 5 Investigating
Task 1 - Boiling Pont
How do you know when a liquid is boiling?
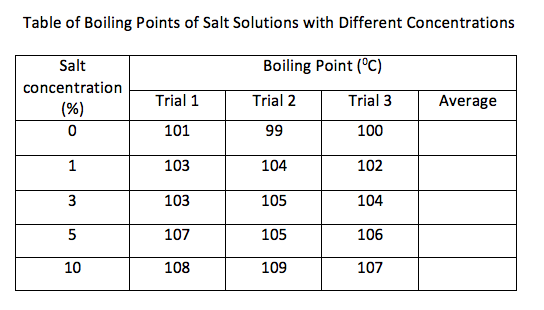
Task 2 Investigation: The Effect of Increasing salt concentration on boiling point
Use the laboratory equipment to investigate the effect of increasing salt concentration on boiling point.
You will set this up this investigation as a Fair Test where you;
You will set this up this investigation as a Fair Test where you;
- identify the independent and dependent variables
- identify variables to be controlled
- suggest the CONTROL, which is the comparison trial.
- write an hypothesis,
- accurately measure,
- conduct three trials within a team,
- record data,
- compare to other teams
- Calculate the average Boiling points for each salt concentartion
- plot average data on a graph
From your boiling point data you can calculate the salt concentration of sea water.
The measurements below are the salt concentrations in a unit called milliSiemens. It is a measure of electrical conductivity. (Salt solutions conduct electricity.)
Calibrate your sea concentration to the salt concentration below.
Now convert the values for salt concentrations in milliSiemens into % concentrations.
The measurements below are the salt concentrations in a unit called milliSiemens. It is a measure of electrical conductivity. (Salt solutions conduct electricity.)
Calibrate your sea concentration to the salt concentration below.
Now convert the values for salt concentrations in milliSiemens into % concentrations.
Discussion
Improvements to the Design of the Investigation
Accuracy of measurement
Improvements to the Design of the Investigation
Accuracy of measurement
- Suggest how to improve accuracy of measurements that were taken.
- Did we select solutions to test that matched the salt concentrations of the land where these plants are to be grown?