Week 8 Electricity
Task 1 Electrostatics Review
|
Science Text page 183 to to 184
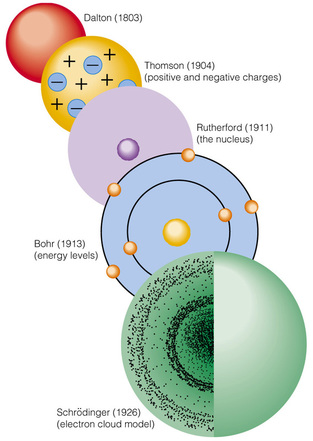
Electric Charge Use the conceptual model of the atom to explain what is meant by "electric charge". Static Electricity
|
Task 2 Electroscopes
|
An electroscope is an instrument that detects whether an object is charged and indicates whether that object has a positive or negative charge.
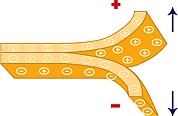
Text page 188 to 189 Electroscopes Follow the instructions to complete this activity by building your own electroscope. Record your observations in a table. Complete the Discussion questions. The button below is a link to instructions so you can make an electroscope at home. Triboelectric charging (a transfer of electrons) occurs when two materials are in contact and are then separated. One material acquires an excess of negative charge and the other an excess of positive charge by transfer of electrons.
|
|
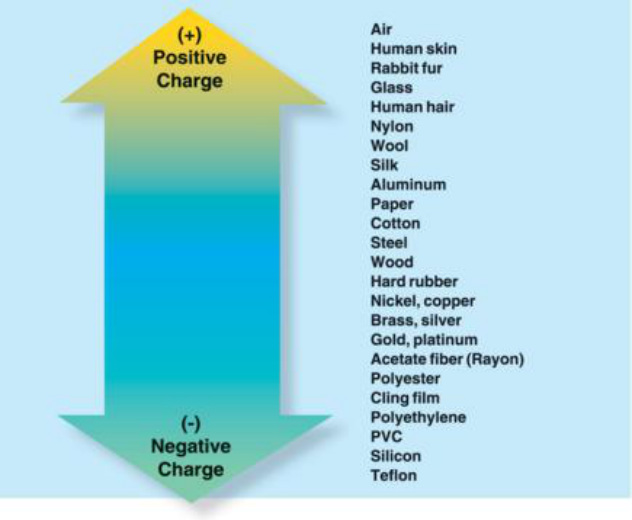
Triboelectric Series is a list that ranks the tendencies of objects to become negative or positive when charged by friction. The image to the left is a Triboelectric Series and shows that, when two objects are rubbed together, the object on top will become positive (lose electrons) while the object on the bottom will become negative (gain electrons). This list also shows a pattern: objects on the top tend to be natural objects (air, human skin, hair) while objects on the bottom tend to be artificial or synthesized (PVC, polyester, teflon).
|
Task 3 View the following
|
|
Static vs Current Electricity
Have you ever seen evidence that "static electricity" can move/flow/transfer?
Text page184
Text page184
A stroke of genius - using the charge of electrons during a chemical reaction to do work!
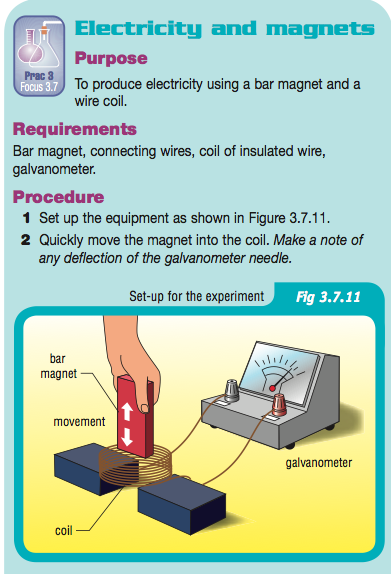
Electrochemical cells and the generator
Michael Faraday was born on 22 September 1791. Faraday was an assistant to Humphry Davy, the English chemist. By age 22 Faraday spent his Friday evenings demonstrating electrical shows to audiences at the Royal Institution. His discoveries of electricity generation and transmission have helped shape the modern world.
http://science.howstuffworks.com/dictionary/famous-scientists/physicists/michael-faraday-info.htm Your practical session today will replicate two of Faraday's experiments that led to the development of the generation of electricity; The electrochemical cell and The generator Reference is SA2 Focus 3.7 page 108 |
Activity 1: Generating electricity with Electrochemical Cells
Before you begin this activity examine the commercial cells. Note the stated voltage. Test this using the multimeter set to record voltage.
Purpose: To make three different electrochemical cells and compare the voltage produced. Requirements per group:
Method:
|
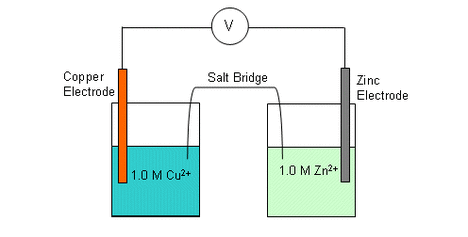
Diagram of an electrochemical cell
Results Table: Cu/Cu2+ // Zn/Zn2+ 0.73 volts Cu/Cu2+ // Pb/Pb2+ 0.49 volts Pb/Pb2+// Zn/Zn2+ 0.29 volts Analysis of results.
|