Week 3 Chemical Changes
Task 1 Changes to Materials
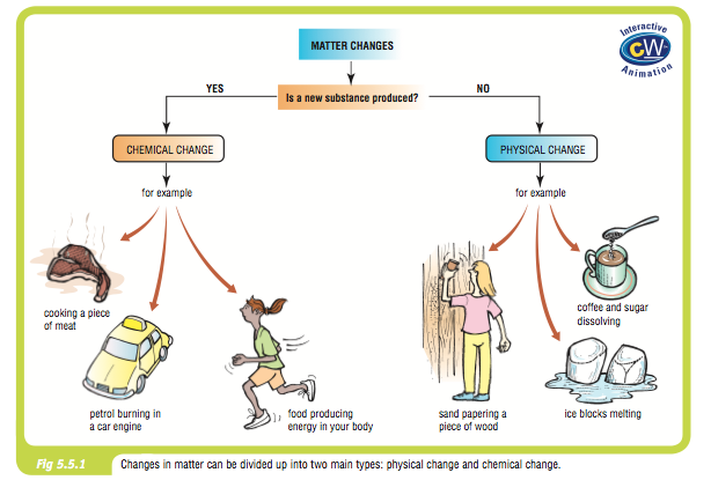
- Use this classification chart as a skeleton to make your own classification chart to classify the features of chemical and physical changes.
- Decide what characteristic is being changed in each of the examples under the heading “Physical Changes"
Task 2 Laboratory Session - Observing and classifying Chemical Changes
Observing Chemical Changes
Science Aspects1 Focus 5.5 Page 242
- Write heading and the Purpose
- Read Requirements, Safety and Procedure
- Draw up a Results Table
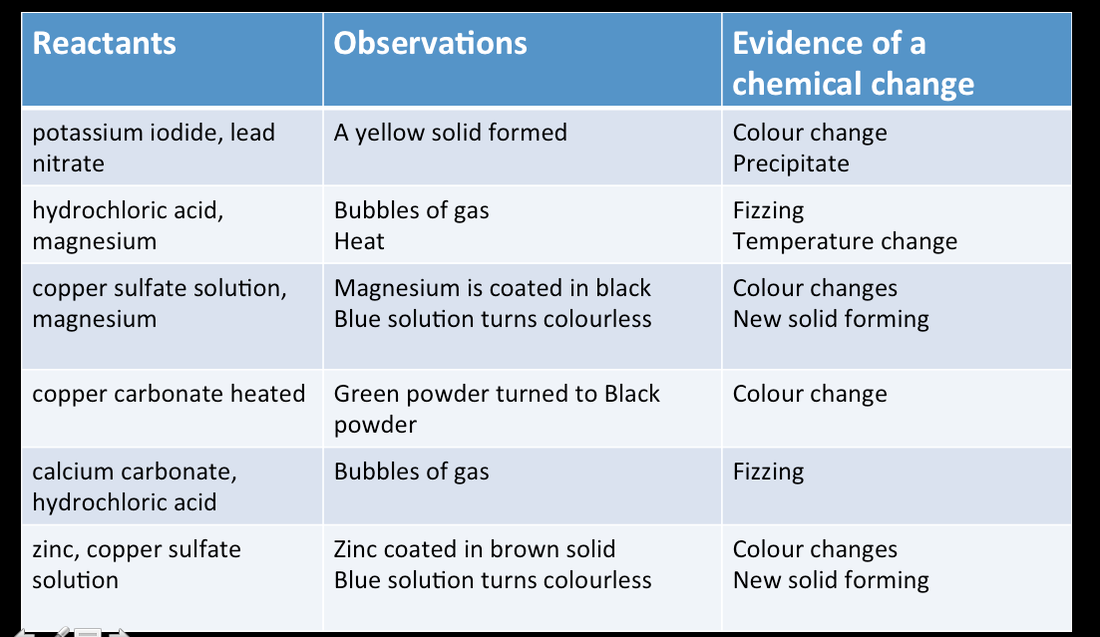
Results
Results filled in
Conclusion
Observe the changes that occur when sodium metal is placed in water
Wear safety glasses and remain away from the reaction vessel.
A general equation to summarise a chemical reaction (chemical change)
reactants ---> products
Write a chemical equation for the following.
Sodium metal was placed in water and universal indicator was added and turned green. Sodium “darted” across the surface of the water, burning with an orange flame.
This means hydrogen gas was given off during the reaction. The solution changed from green to purple, indicating that sodium hydroxide had formed.
Sodium metal was placed in water and universal indicator was added and turned green. Sodium “darted” across the surface of the water, burning with an orange flame.
This means hydrogen gas was given off during the reaction. The solution changed from green to purple, indicating that sodium hydroxide had formed.
Electrolysis of water using a Hoffman Voltameter
An electric current is passed through water. Two colourless gases are produced. One gas at one electrode has twice the volume of the other gas.
One gas was predicted, tested and confirmed to be oxygen. The gas with twice the volume was predicted, tested and found to be hydrogen.
One gas was predicted, tested and confirmed to be oxygen. The gas with twice the volume was predicted, tested and found to be hydrogen.