Week 4 Electron arrangement and Ion Formation
Task 1 - Electron arrangement in the atoms of the first twenty elements of the periodic table
Bohr Model of atoms
As you know there are so many atoms, elements, molecules, ions and compounds to learn about. If we look for patterns as scientists do, then we can organise our thinking.
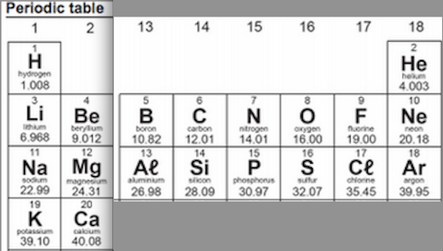
Fold your periodic table so that it looks like this;
Fold your periodic table so that it looks like this;
The blank grid looks like the table above and goes on the Right-side page (input)
On your grid write the symbol for the atom and its atomic number in the top of each cell.
The rules for filling energy levels are,
Up to;
On your grid write the symbol for the atom and its atomic number in the top of each cell.
- Recall the meaning of atomic number and note this. Explain the significance of the atomic number to both the atom and the arrangement of the periodic table.
- Recall the meaning of the mass number. What is the significance of the mass number in regard to atomic structure? Is there any trend or pattern that you can identify in regard to mass number and the periodic table?
The rules for filling energy levels are,
Up to;
- two electrons can be held in the first energy level
- eight electrons can be held in energy levels two and three.
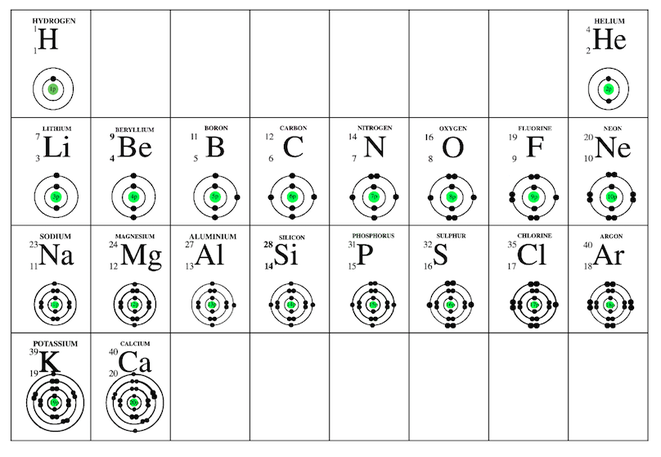
Electron arrangement of the first twenty elements of the periodic table.
Right-side page (Input)
Left-side page (Output)
The circles that you have drawn represent energy levels of the electrons.
The circles that you have drawn represent energy levels of the electrons.
- Describe the similarities of atoms across Period 1, across Period 2, across Period 3, across Period 4.
- Describe the similarities of atoms down Group 1, down Group 2, down Group 13, down Group 14, down Group 15, down Group 16, down Group 17, down Group 18.
- Describe what happens to the diameter of atoms down a group. Explain why this occurs.
- Describe what happens to the diameter of atoms across a period. Explain why this occurs.
Electron Configuration
Drawings showing electron arrangement are useful for us to visualise the atoms and understand some trends. They are cumbersome to draw.
A shorthand for these drawings is to use a notation called electron configuration.
The electron configuration of a calcium atom is written like this; 2, 8, 8, 2
Compare this notation to the drawings of the atoms on the periodic table
A shorthand for these drawings is to use a notation called electron configuration.
The electron configuration of a calcium atom is written like this; 2, 8, 8, 2
Compare this notation to the drawings of the atoms on the periodic table
- What do the numbers represent?
- What do the commas represent?
- What does the space, that the number is in, represent?
- How does information tell us where each element is found on the periodic table?
- Complete writing the electron configurations for the first twenty elements.
Another Video: Trends in the Periodic Table
Trends in the periodic table can reveal important properties and characteristics of the different elements. Here is more information on trends in the periodic table.
http://video.about.com/chemistry/Video--Trends-in-the-Periodic-Table.htm#vdTrn
Trends in the periodic table can reveal important properties and characteristics of the different elements. Here is more information on trends in the periodic table.
http://video.about.com/chemistry/Video--Trends-in-the-Periodic-Table.htm#vdTrn
Task 2 Indirect observation of electron shells
Text page 12
Right-hand page (Input)
Purpose: To observe coloured light produced when electrons jump from one electron shell to another.
Method: You teacher will demonstrate the method.
Results: Draw a results table like the one on page 12
Discussion: Record point 1 information under the results table.
Left-side page (Output)
Text page 12: Answer the discussion questions 1 and 2
Purpose: To observe coloured light produced when electrons jump from one electron shell to another.
Method: You teacher will demonstrate the method.
Results: Draw a results table like the one on page 12
Discussion: Record point 1 information under the results table.
Left-side page (Output)
Text page 12: Answer the discussion questions 1 and 2
Task 3 Consolidation of understandings of Atomic structure
Text work
Read pages 2 to 4 "Atoms in Elements and Compounds" an introduction. We will clarify these concepts later.
On a new Right-side (Input) page Write the Heading "Inside Atoms"
Read pages 5 to 7.
Write the sub-heading "Remembering"
Answer the questions 1 to 5
Write the sub-heading "Understanding"
Answer the questions 6 to 11
Flip to the Left-side page (output) to answer the higher-order-thinking questions
Write the sub-heading "Applying"
Answer the questions 10 to 14
Write the sub-heading "Analysing"
Answer the questions 15 to 16
You have completed all the remaining questions as an inquiry in previous lessons.
Read pages 2 to 4 "Atoms in Elements and Compounds" an introduction. We will clarify these concepts later.
On a new Right-side (Input) page Write the Heading "Inside Atoms"
Read pages 5 to 7.
Write the sub-heading "Remembering"
Answer the questions 1 to 5
Write the sub-heading "Understanding"
Answer the questions 6 to 11
Flip to the Left-side page (output) to answer the higher-order-thinking questions
Write the sub-heading "Applying"
Answer the questions 10 to 14
Write the sub-heading "Analysing"
Answer the questions 15 to 16
You have completed all the remaining questions as an inquiry in previous lessons.
Task 4 Formation of ions
Lets examine the evidence for formation of ions from atoms.
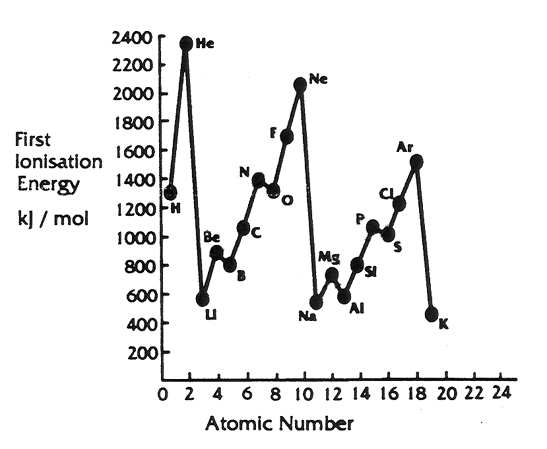
Below is a graph of First Ionisation Energy plotted against Atomic Number. You know that atomic number refers to specific atoms. Their symbols appear on the dots on the graph.
The first ionisation energy of an atom describes the quantity of energy required to remove the first outer electron from the outer-most energy shell of an atom in the gaseous state.
The graph shows data collected when the different elements, as gases, are forced to lose one of their outermost electrons.
Glue this graph to your Right-side page (Input) with the heading;
Formation of ions.
Below is a graph of First Ionisation Energy plotted against Atomic Number. You know that atomic number refers to specific atoms. Their symbols appear on the dots on the graph.
The first ionisation energy of an atom describes the quantity of energy required to remove the first outer electron from the outer-most energy shell of an atom in the gaseous state.
The graph shows data collected when the different elements, as gases, are forced to lose one of their outermost electrons.
Glue this graph to your Right-side page (Input) with the heading;
Formation of ions.
Analysis of the graph showing the relationship between ionisation energy and atomic number.
- Predict the First Ionisation Energy of Calcium.
- Describe the ease of electron removal from atoms of elements across Periods 1, 2, and 3 of the Periodic Table.
- You know that atoms have a neutral charge. Explain why this is so.
- When atoms lose or gain electrons they become charged and are called IONS. Positively charged ions are called CATIONS. Negatively charged ions are called ANIONS.
- If an atom such as sodium loses one electron what is the magnitude and value of the charge on the ion? Write the electron configuration of the sodium ion.
- If an atom such as magnesium loses two of its outer electrons what is the magnitude and value of the charge on the ion? Write the electron configuration of the magnesium ion.
- If an atom such as aluminium loses three of its outer electrons what is the magnitude and value of the charge on the ion? Write the electron configuration of the aluminium ion.
- Compare the electron configuration of the three metal ions. What do you notice?
- Describe another way of stating this.
- If an atom such as chlorine gains one electron what is the magnitude and value of the charge on the ion? Write the electron configuration of the chloride ion.
- If an atom such as oxygen gains two of its outer electrons what is the magnitude and value of the charge on the ion? Write the electron configuration of the magnesium ion.
- If an atom such as nitrogen gains three of its outer electrons what is the magnitude and value of the charge on the ion? Write the electron configuration of the nitride ion.
- Compare the electron configuration of the three non-metal ions. What do you notice?
- Describe another way of stating the observation in "13" above.
- Compare how we name metal ions (cations) and non-metal ions (anions).
Lewis Dot Diagrams
Lewis dot diagrams of atoms show only the valence electrons.
It is the valence electrons that are involved in chemical reactions.
Show electrons as a dot.
Electrons repel each other so spread the dots evenly around the atom... but when you get more than four electrons start making pairs of electron dots.
Draw Lewis Dot diagrams of the first twenty atoms of the Periodic Table. Arrange them in the matrix of Groups and Periods.
Describe the pattern down the Groups.
Describe the pattern across the Periods.
It is the valence electrons that are involved in chemical reactions.
Show electrons as a dot.
Electrons repel each other so spread the dots evenly around the atom... but when you get more than four electrons start making pairs of electron dots.
Draw Lewis Dot diagrams of the first twenty atoms of the Periodic Table. Arrange them in the matrix of Groups and Periods.
Describe the pattern down the Groups.
Describe the pattern across the Periods.
Lewis dot diagrams help us understand how atoms bond together.
Task 5 - Consolidation of "Ion Formation"
Ions form when an atom _____________________ or _________________________ its _______________________ electrons.
If an atom loses valence electrons then a ____________________________ ion forms. A positive ion is called a __________________ .
Atoms on the left side of the periodic table, or ________________ atoms with up to ___________________ valence electrons become cations.
If an atom gains valence electrons then a ____________________________ ion forms. A negative ion is called an __________________ .
Atoms on the right side of the periodic table, or ___________________ atoms with from ____________________ to ___________________ valence electrons become anions.
Both cations and anions have ________________________ outer shells of electrons, or _______________ gas electron configuration.
The ____________________________________ force between the positively charged __________________ and the negatively charged ______________________ form an ____________________ bond. This is a very strong bond.
If an atom loses valence electrons then a ____________________________ ion forms. A positive ion is called a __________________ .
Atoms on the left side of the periodic table, or ________________ atoms with up to ___________________ valence electrons become cations.
If an atom gains valence electrons then a ____________________________ ion forms. A negative ion is called an __________________ .
Atoms on the right side of the periodic table, or ___________________ atoms with from ____________________ to ___________________ valence electrons become anions.
Both cations and anions have ________________________ outer shells of electrons, or _______________ gas electron configuration.
The ____________________________________ force between the positively charged __________________ and the negatively charged ______________________ form an ____________________ bond. This is a very strong bond.
Consolidation of "Ion Formation"
Text Chapter 1.2 pages 13 to 15
Make notes on a new Right-side page (input) under the subheadings;
Atoms and Ions
Cations
Anions
Ionic Compounds
Chemical Names and formulae
Atoms and Ions
Cations
Anions
Ionic Compounds
Chemical Names and formulae